Introduction to Extracellular Vesicles (EVs):
Extracellular vesicles (EVs) are lipid bilayer membrane bound structures in extracellular fluids, which are secreted by all types of cells (either in normal or pathogenic conditions). They are naturally released from the cell that are delimited by a lipid bilayer and cannot replicate. EVs are complex signals which can target recipient cells through surface molecules and RNA. The major role of EVs is in intracellular communications, and are involved in transport of biological signals to regulate diverse cellular function.
Once recognized or internalized, EVs are able to modify the physiological state of the recipient cell. All living cells have the capacity to release EVs, including mammalian cells, fungi, plants, protozoa and prokaryotes like bacteria.
EVs can be found everywhere. Some of the most studied EV samples are plasma/serum, urine, feaces, cerebrospinal fluid (CSF), breast milk, and amniotic fluid.
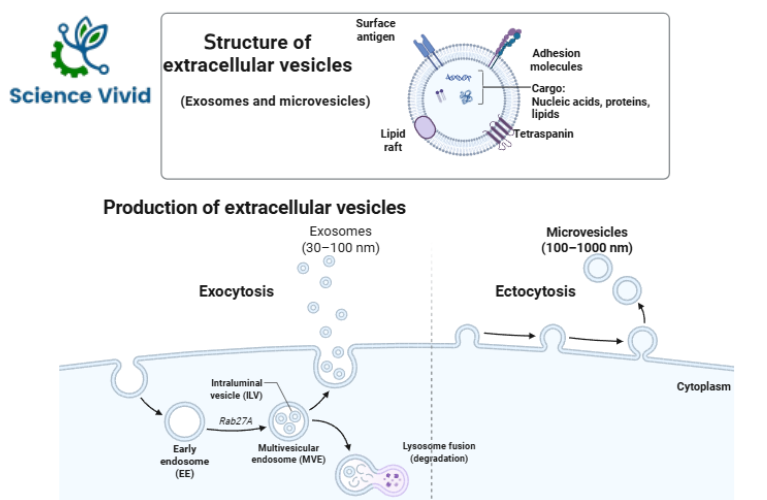
Fig: Structure and prodcution of Extracellular Vesicles (EVs)
Types of Extracellular Vesicles (EVs):
Each eukaryotic cell releases a heterogeneous population of EVs, with three broad categories of EVs depending on their biogenesis mechanism. Historically named exosomes, microvesicles, and apoptotic bodies. The most studied EV type is exosomes.
Exosomes
- They have a defined biogenesis mechanism. They form by inward budding of the limiting membrane of early endosomes, which mature into multivesicular bodies and are secreted when these compartments fuse with the plasma membrane and release their contents into the extracellular space.
- Exosomes are typically less than 160 nm in diameter and small at ~40–60 nm (in diameter) that originate from exocytosis of multivesicular bodies (MVBs).
- They contain proteins, nucleic acids (mRNA, miRNA and other non-coding RNAs) and lipids.
- Intercellular communication is also the major function of exosomes.
- Because of the defined biogenesis mechanism, exosomes are enriched in endosome-derived markers that can be used to validate the presence of this type of EVs in a sample.
- Despite having the simplified origin, the exosome population released from a single cell can be very heterogeneous and will adapt depending on the stimulus the cells are exposed to. There are different exosome subtypes that can have more specific
- Size range and molecular composition. It is well established that exosome roles are very diverse, not only in pathological conditions but in normal cellular activities. Exosomes are associated with immune responses, wound healing, pregnancy, viral pathogenicity, cardiovascular diseases (CVD), central nervous system (CNS) related diseases, and cancer progression.
Microvesicles or ectosome
- The second most studied EV. It is formed by budding of the plasma membrane, a releasing process that is driven by calcium-dependent signaling. It involves activity of several enzymes, cytoskeleton remodeling, and externalization of specific phospholipids. Microvesicles are shed under normal conditions and their release increases with various stimuli and pathological settings.
- It falls in a diverse range of sizes, from 100 to 1000 nm in diameter and may also be called ectosomes or microparticles. It can package a variety of distinct biomolecules, like lipids, genetic material and proteins such as transcription factors, cytokines, and growth factors.
- Markers for microvesicles include surface molecules from parental cells and integrins or CD40 among others.
- They contain proteins, nucleic acids (mRNA, miRNA and other non-coding RNAs) and lipids.
- Intercellular communication is the major function of microvesicles.
Apoptotic bodies
They are EVs specifically generated through apoptotic cell disassembly. They are large EVs, typically 500 nm to 5 µm in diameter. Apoptotic bodies can harbor DNA, RNA and proteins; therefore, they can mediate intercellular communication through the transport of these biomolecules and may contribute to the development of various disease states. There are some markers that can help identify apoptotic bodies, like histones, annexin V and the presence of organelles. They facilitate phagocytosis.
Prokaryotic EVs
These are bacterial EVs. EVs differ from eukaryotic cells as they have a faster growth pace and don’t have organelles. Therefore, all molecular machinery and processes occur free in cytoplasm. Bacteria are smaller than eukaryotic cells, ranging from 0.2–3 µm, and have complex and different cell wall compositions. These unique growth and structural characteristics have resulted that EVs can be generated by different mechanisms,
- Depending on their cell wall composition:
- Exposure to environmental stimulus.
Bacterial EVs have been described with sizes between 50 and 300 nm and may contain a diverse cocktail of molecular cargo, like DNA, RNA, proteins, lipids, and metabolites.
Isolation Methods Extracellular vesicles (EVs):
- To isolate EVS for clinical research or therapeutic development, EVs must be separated from contaminants, other particles, and sub cellular particles.
- Though ultracentrifugation-based techniques are still the gold standard for isolating exosomes, alternative techniques have been developed to overcome ultracentrifugation’s drawbacks. These alternative techniques have been developed based on exosome precipitation, immunoaffinity capture, and isolation by size; nevertheless, even these techniques are unable to isolate exosomes entirely and usually produce complicated mixes of EVs and other extracellular space components.
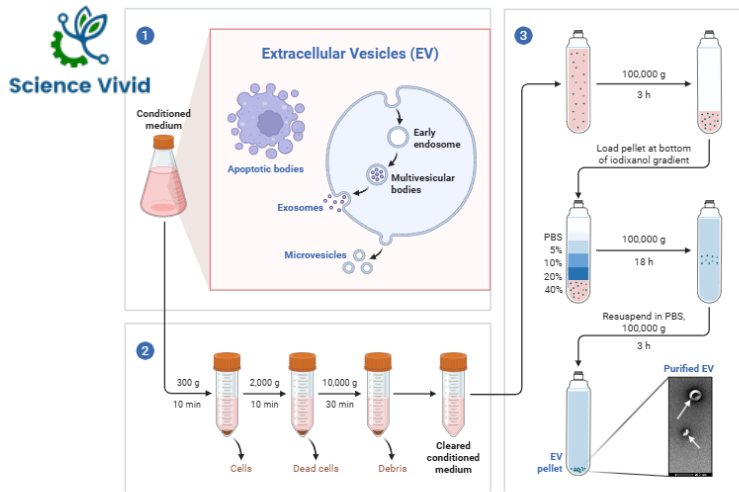
Fig: Isolation of Extracellular vesicles (EVs)
1) During regular cell activity, cells release extracellular vesicles (EV).
2) Conditioned culture media is collected for EV separation, and significant impurities are eliminated by a series of low-speed centrifugations.
3) A 100K pellet is put onto the bottom of an iodixanol gradient after the cleared supernatant has been condensed by ultracentrifugation. As EVs rise, EV-enriched fractions are gathered and ground into pellets. The final sample is free of impurities and high in EVs.
Extracellular vesicles as tools and targets in therapy for diseases:
- EVs act as a potential diagnostic marker for several types of diseases such as cancer. Because they play a major role in cellular communication, they are also used as a therapeutic agent,
- Because EVs naturally serve as transporters, they can potentially be good vectors for delivering exogenously loaded drugs.
- EVs can be used for regenerative medicine, cancer, and infectious diseases as both a diagnostic and therapeutic tool.
References:
- Zhao, Z., Wijerathne, H., Godwin, A.K. and Soper, S.A., 2021. Isolation and analysis methods of extracellular vesicles (EVs). Extracellular vesicles and circulating nucleic acids, 2(1), p.80.
- Abels, E.R. and Breakefield, X.O., 2016. Introduction to extracellular vesicles: biogenesis, RNA cargo selection, content, release, and uptake. Cellular and molecular neurobiology, 36(3), pp.301-312.
- Di Bella, M.A., 2022. Overview and update on extracellular vesicles: considerations on exosomes and their application in modern medicine. Biology, 11(6), p.804.
