Introduction:
Isoelectric point (pI) is the pH at which the net electric charge on a molecule/protein is zero. At its pI, the molecule/protein is electrically neutral that the molecule at its pI exists as a zwitterion with an equal number of positive charges (from protonated amino groups) and negative charges (from deprotonated carboxyl groups).
At pH values below and above the isoelectric point, the molecule will bear a net positive or net negative charge, respectively.
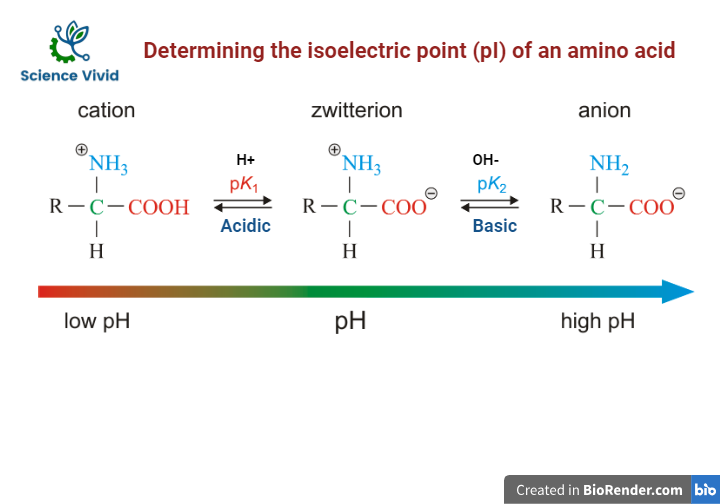
Fig: Determining the isoelectric point (pI) of an amino acid
It can be easily calculated using the formula, pI = pKa1 + pKa2/ 2 for molecules with two ionizable groups, while for molecules with more than two ionizable groups, pI is the average of two pKa values that are close to each other.
Determine the pKa value(s):
Amino acids can have multiple ionizable groups, such as the amino group (-NH2) and the carboxyl group (-COOH). Some amino acids also have additional ionizable side chains, such as the imidazole group in histidine (His).
- For the amino group, the pKa is around 9-10.
- For the carboxyl group, the pKa is around 2-3.
Materials Required:
- Amino acid sample
- Strong acid (e.g., HCl) or strong base (e.g., NaOH) solution for titration
- pH meter or indicator
- Burette or pipette for accurate volume measurement
- Beakers or flasks for mixing solutions
- Stirring rod
Procedure:
Preparation of Amino Acid Solution
Prepare 0.1M solution of given amino acid in a distilled water and ensure the solution is thoroughly mixed and fully dissolved accordingly.
Titration
Firstly, dispense 10 ml of freshly prepared 0.1M amino acid solution in a beaker and begin the titration by slowly adding 1ml of freshly prepared 0.1M HCL to the amino acid solution while stirring continuously. With the addition of the acid in the amnio acid solution, the pH of the amnio solution will decline. Record the pH of the solution after each incremental addition of the acid solution and ensure accurate pH measurements using a calibrated pH meter. Subsequently, 1ml of 0.1M HCL will be continuously added to the amino acid solution and repeat the procedure until a consistent pH value is recorded.
Secondly, pour 10 ml of another fresh 0.1M amino acid solution into a beaker and begin the titration by slowly adding 1ml of freshly prepared 0.1M NaOH to the amino acid solution while stirring continuously. With the addition of the base in the amnio acid solution, the pH of the amnio solution will rise. Record the pH of the solution after each incremental addition of the base solution and ensure accurate pH measurements. Subsequently, 1ml of 0.1M NaOH will be continuously added to the amino acid solution and repeat the procedure until a consistent pH value is recorded.
Analysis
a. Plot a graph of pH versus volume of titrant added and identify the point of inflection on the titration curve, which corresponds to the pKa value of the amino acid. Determine the pKa value by extrapolating or interpolating the pH value at the midpoint of the buffering region.
Calculate the pI
The pI of an amino acid is the pH at which it carries no net electrical charge. This occurs when the number of positive charges (from protonated amino groups) equals the number of negative charges (from deprotonated carboxyl groups and any side chain groups). To estimate the pI, consider the following:
If the amino acid has only one ionizable group (e.g., glycine), the pI will be the average of its pKa value and the pKa of the opposing ionizable group. For example, for glycine (pKa values around 9-10 for the amino group and 2-3 for the carboxyl group), the pI would be around (9 + 2) / 2 = 5.5.
If the amino acid has multiple ionizable groups (e.g., histidine with an additional side chain group), estimate the pI based on the pKa values of all relevant ionizable groups. Calculate PI using the formula, pI = pKa1 + pKa2/ 2.
Applications
- Protein purification
- Biomedical research
- Drug delivery
- Environmental monitoring
- Protein Characterization
- Understanding Protein-Protein Interactions
