Introduction:
It is also known as glyoxylate shunt (GS), was identified by Kornberg and Krebs in 1957, explaining how organisms could grow on acetate as the sole carbon source (Kornberg and Krebs, 1957).
The glyoxylate cycle, an anabolic pathway, is a variation of the tricarboxylic acid cycle, is an anabolic pathway occurring in plants, bacteria, protists, and fungi that allows utilization of two carbons compounds in the absence of glucose. However, it is generally not present in human and animal tissue.
Steps:
The key feature of the glyoxylate cycle is the conversion of isocitrate to glyoxylate and succinate without the two decarboxylation steps that occur in the TCA cycle. The glyoxylate cycle involves the following key enzymatic steps:
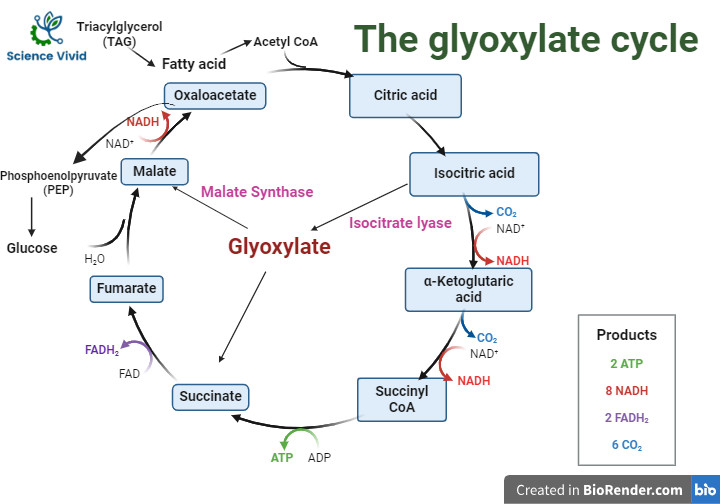
Fig: The glyoxylate cycle/Shunt (GS)
Isocitrate lyase: This enzyme catalyzes the conversion of isocitrate to glyoxylate and succinate. Unlike the isocitrate dehydrogenase in the TCA cycle, isocitrate lyase does not decarboxylate isocitrate.
Malate synthase: In the second step, glyoxylate reacts with acetyl-CoA to form malate. This reaction is catalyzed by malate synthase.
Biochemical Importance:
- The glyoxylate cycle is particularly important for organisms that utilize fatty acids as a primary carbon source, as it allows them to generate glucose from acetyl-CoA derived from the breakdown of fatty acids.
- Plants often employ the glyoxylate cycle in germinating seeds where fatty acids stored in oil serve as an energy source for seedling growth.
Significance:
Enzyme/Metabolic Engineering
The glyoxylate cycle has been extensively studied and explored concerning the enzymology and metabolic regulation for the bioproduction of required of organic acids, amino acids, and fatty acid-related in the field of metabolic engineering particularly for Escherichia coli and the pathogenic bacterium Mycobacterium tuberculosis.
Role of glyoxylate cycle in pathogenesis
Fungi are eukaryotic organisms that can cause a wide variety of infectious diseases in humans, from acute surface infections to potentially fatal systemic infections. C. albicans and C. glabrata that had been absorbed by macrophages showed substantial metabolic reprogramming, indicating food deprivation. Furthermore, the involvement of the glyoxylate cycle in the pathogens of certain bacteria, such as Mycobacterium tuberculosis, is being investigated.
The effects include upregulation of genes that encode key metabolic enzymes in three interconnected anabolic pathways for alternative carbon metabolism, i.e., gluconeogenesis (PCK1 and FBP1), glyoxylate cycle (ICL1 and MLS1), and β-oxidation of fatty acids (FOX2, POT1, and POX1), as well as downregulation of glycolysis-related genes.
The activation of these important enzyme genes indicates that the microenvironment within macrophages lacks glucose; nonetheless, confined C. glabrata cells can still use endogenous resources such as alternate carbon sources for growth and survival through glyoxylate cycle.
