Introduction to Pseudomonas aeruginosa:
The gram-negative, opportunistic bacterium Pseudomonas aeruginosa is now one of the leading causes of infections in hospitals and the population globally. Although it occurs naturally in soil, water, and damp habitats, it can pose a major threat to susceptible hosts. Individuals with compromised immune systems—such as those receiving chemotherapy, people living with HIV/AIDS, burn patients, and particularly those suffering from long-term lung conditions like cystic fibrosis—are particularly vulnerable.
The adaptability of the organism—which includes several virulence factors, protective biofilm formation, quorum sensing for communication, and both innate and acquired resistance to a broad spectrum of antibiotics—makes it clinically significant. These characteristics make P. aeruginosa infections difficult to cure, frequently persistent, and linked to high rates of morbidity and mortality.
Infection Routes and Initial Colonization:
Infection begins with entrance into the host and tissue colonization. P. aeruginosa can enter the body through skin injuries, such as burns or wounds, the respiratory system, particularly in patients on a ventilator, the urinary system (via the use of catheters).
Surface features like pili and flagella, which mediate adherence to epithelial cells, aid colonization once inside. Following adhesion, the bacterium creates structured communities called biofilms, which are covered with extracellular matrix that it produces on its own. Biofilms act as a barrier between the host immune system and antibiotics. Patients with cystic fibrosis are particularly affected because their thick mucus creates the perfect environment for biofilm formation, which can result in chronic lung infections that can linger for decades.
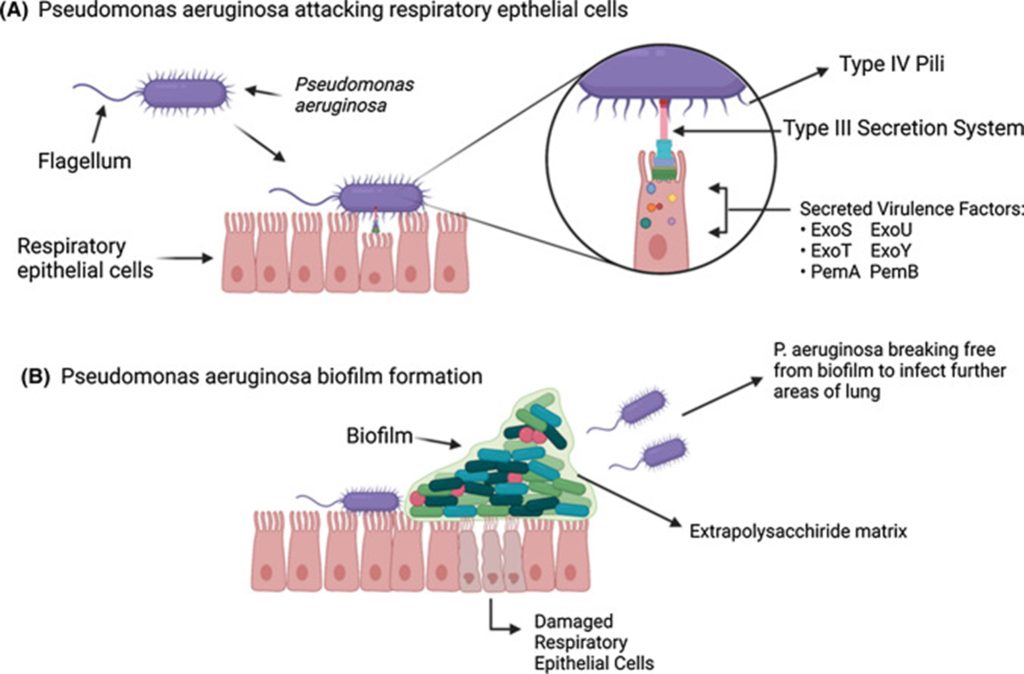
Fig: Pathogenesis of Pseudomonas aeruginosa causing human infection
Major Virulence Factors of P. aeruginosa:
Lipopolysaccharide (LPS)
P. aeruginosa’s lipopolysaccharide (LPS), a crucial component of the outer membrane, is one of its main virulence factors. LPS has both pathogenic and structural functions.
Lipid A
Lipid A Component initiates the NF-κB pathway by binding to host receptors such MD2 and CD14. Systemic inflammation is the outcome of this signaling cascade.
Exotoxin A (ETA)
Exotoxin A (ETA) is one of the most powerful virulence factors. ETA is composed of two structural pieces. Fragment B permits access by attaching to particular host cell receptors. Fragment A stops protein synthesis by altering elongation factor-2 (EF-2) through ADP-ribosylation.
Lectins LecA and LecB
In addition to killing host cells, this activity causes significant tissue damage. In addition to its cytotoxicity, ETA helps depress the immune system, which makes it easier for P. aeruginosa to colonize and invade. Lectins LecA and LecB P. aeruginosa also generates lectins, which are proteins that bind carbohydrates and have two functions in Pathogenicity.
- Bind to sugars on the surface of epithelial cells to aid in ETA absorption.
- Cause cytotoxicity by directly harming epithelial cells.
LecA and LecB not only facilitate bacterial attachment but also amplify both the direct cellular injury and toxin-related damage, thereby intensifying the overall severity of P. aeruginosa infections.
Type III Secretion System (T3SS) and Effector Proteins:
One of the most striking virulence mechanisms of this bacterium is the Type III Secretion System (T3SS). This apparatus functions like a microscopic syringe, enabling the direct injection of bacterial effector proteins into host cells.
The principal effectors delivered through T3SS include:
- ExoS, which disrupts the actin cytoskeleton and damages lung tissue. Its ADP-ribosyltransferase activity targets Ras and Rab GTPases, impairing DNA synthesis and weakening cellular integrity.
- ExoU, a highly cytotoxic factor that works alongside ExoT to destroy epithelial cells. By promoting the leakage of pro-inflammatory cytokines into the bloodstream, it plays a critical role in the development of septic shock.
- ExoY, which elevates intracellular cAMP concentrations, resulting in abnormal actin remodeling and interference with host cell signaling pathways.
Together, these effectors orchestrate immune evasion, extensive tissue destruction, and programmed host cell death.
Quorum Sensing (QS):
In addition to T3SS, P. aeruginosa relies on a highly advanced communication network known as quorum sensing (QS). This system allows the bacterial population to regulate its behavior in a density-dependent manner. QS operates through signaling molecules called acyl-homoserine lactones (AHLs) and their associated regulatory proteins. Another crucial signaling molecule, the Pseudomonas quinolone signal (PQS), governs the expression of genes involved in toxin production, enzyme secretion, and tissue invasion.
Through QS, P. aeruginosa ensures that virulence factors are expressed only when bacterial numbers reach a critical threshold, thereby maximizing their impact against host defenses.
A particularly important role of QS is in the shift from acute infections to chronic, persistent states. In conditions like cystic fibrosis, the pathogen transitions into a mucoid form characterized by excessive production of alginate, a protective polysaccharide. This alginate shields the bacterium from pulmonary clearance mechanisms, phagocytosis, antibodies, and complement activity, while also increasing resistance to antimicrobial therapy.
One of the hallmark features of P. aeruginosa is its ability to establish long-term infections without systemic spread. In cystic fibrosis patients, for instance, the infection may endure for decades. The dominance of mucoid colonies and alginate overproduction provide a survival advantage, allowing the pathogen to persist and thrive despite continuous immune pressure and therapeutic intervention.
If local barriers are breached, P. aeruginosa can invade the bloodstream, leading to systemic infection. Key mediators include:
- Elastases (LasA, LasB) and alkaline protease, which degrade tissues and enable spread.
- ExoS, which destroys lung tissue and aids dissemination.
- Lipid A, which drives the inflammatory cascade characteristic of sepsis.
- Dissemination often results in septicemia, septic shock, and multi-organ failure, all of which carry a high mortality risk.
Clinical Implications:
P. aeruginosa is a leading cause of hospital-acquired infections, including:
- Ventilator-associated pneumonia.
- Urinary tract infections.
- Wound and burn infections.
- Bloodstream infections.
Its natural resistance to many antibiotics, combined with biofilm formation, makes treatment particularly challenging. Multidrug-resistant (MDR) and extensively drug-resistant (XDR) strains are increasingly reported worldwide.
Current Therapeutic Challenges:
- Requires potent antipseudomonal antibiotics (e.g., carbapenems)
- Often necessitates combination therapy due to resistance.
- New strategies being investigated include quorum sensing inhibitors, biofilm-disrupting agents, and vaccines.
Reference:
- Reynolds, D., & Kollef, M. (2021). The Epidemiology and Pathogenesis and Treatment of Pseudomonas aeruginosa Infections: An Update. Drugs, 81(18), 2117–2131. https://doi.org/10.1007/s40265-021-01635-6
